Confirm Your Position in the Fastest-Growing CNS Modality
With Biogen’s acquisition of Apellis, Novartis’ acquisition of Avidity, and Roche’s $2bn deal with Manifold Bio, the momentum in the field is unmistakable.
As CNS pipelines expand and competition intensifies, now is an optimal moment to position your organization at the forefront of oligonucleotide innovation. Unlock new commercial opportunities with a highly targeted global audience, building on five years of proven momentum.
As the field evolves, so do the challenges. The rise of second-generation BBB shuttle strategies beyond TfR1 is increasing demand for advanced delivery technologies. At the same time, increasingly complex, conjugated oligos require sophisticated synthesis, optimization, and manufacturing capabilities.
With more companies entering the space, there is a growing need for trusted partners and specialist vendors who can support the next wave of innovation.

Key Interest Areas
Attendees are actively looking for service and solution providers with capabilities in but not limited to:
CNS Delivery & BBB Shuttle Technologies
Solutions for targeted CNS delivery, including second-generation BBB shuttles for receptor-mediated transport and alternative targeting strategies beyond TFR1 & CD98.
Chemistry & Conjugation Platforms
Capabilities in chemical modification and conjugation, including peptide, lipid, carbohydrate, and antibody conjugates, as well as linker optimization to improve delivery, stability and safety.
Oligonucleotide Synthesis
Custom oligonucleotide synthesis with reliable, advanced sequence and chemical modifications to support early discovery and lead optimization.
Regulatory & Clinical Strategy
Support with regulatory pathways, IND-enabling studies, and clinical development strategies for CNS oligonucleotide therapeutics.
Preclinical CNS Models & CRO Services
CNS-relevant preclinical models such as rodent, NHP, organoid, lab-on-chip to assess PK/PD, long term neurotoxicity and target engagement.
CMC, Scale-Up & GMP Manufacturing from CDMOs
Development and manufacturing of oligonucleotides, including process development, scale-up, and GMP production as more biopharma enter clinical trials.
Analytical Characterization & Quality Control
Analytical and purification services, including impurity profiling, stability testing, and quality control to support regulatory readiness and drug product integrity.
Hear What Our Past Sponsors Have to Say

The meeting was very interactive. The balance between the presentation and the roundtable discussion was really good. I learned a lot and made valuable connections as well.”
Setsu Endoh- Yamagami, Senior Scientist, FUJIFILM
Audience Composition
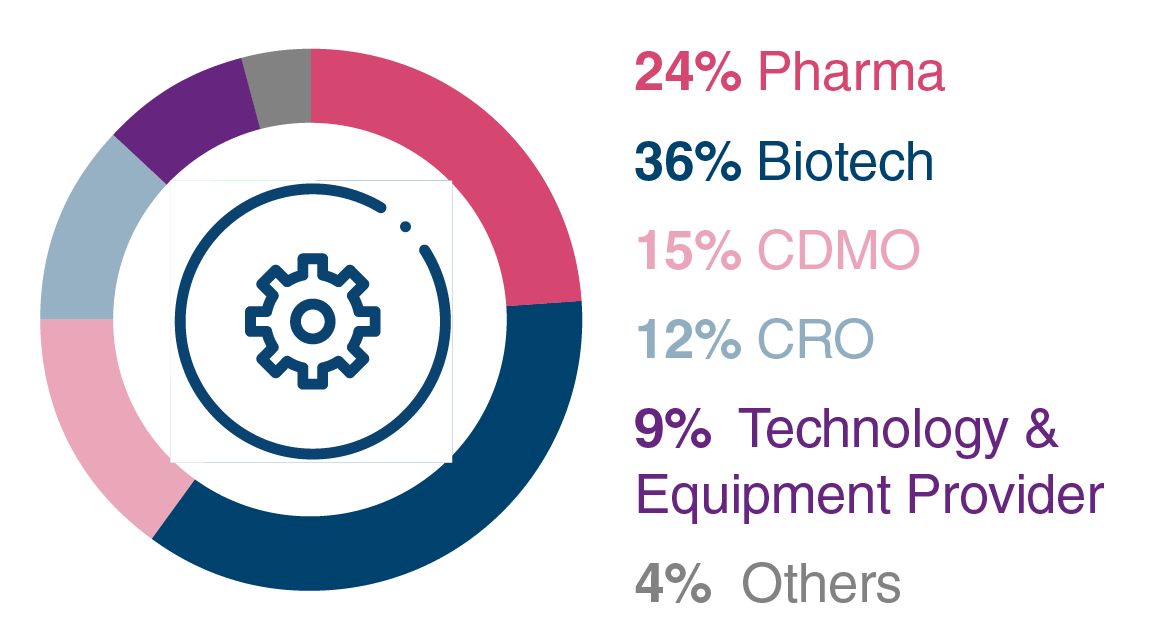
Company Type
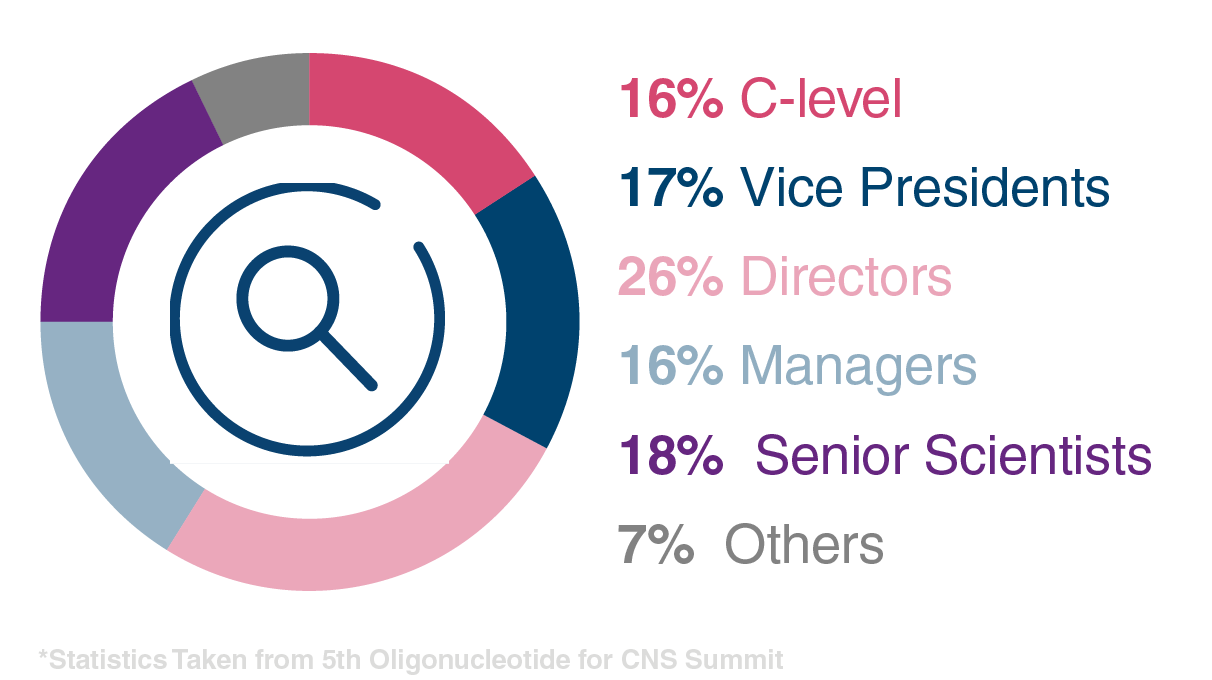
Attendee Seniority
Attending Companies Include








Available Inventory
We have a range of inventory from standard booth and speaking slots to more bespoke deliverables such as lanyards, exclusive dinners and tours to help elevate your brand.








Get in Touch
Take advantage of our bespoke sponsorship opportunities to achieve your commercial goals. Email us if you would like to get involved and discuss a bespoke package suited to your needs.

Katie Orchard
Partnerships Director



